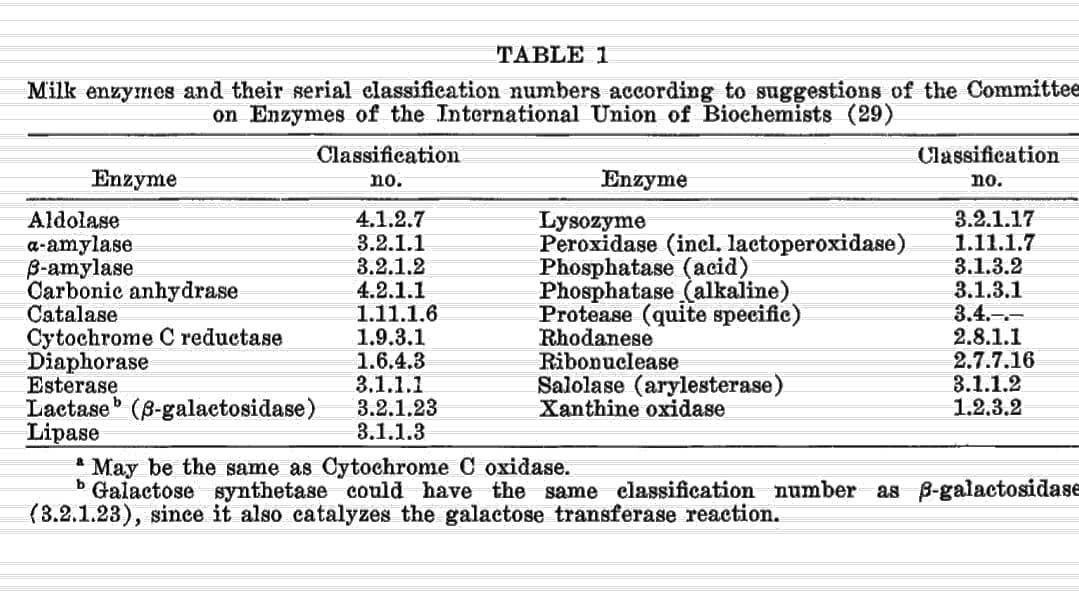
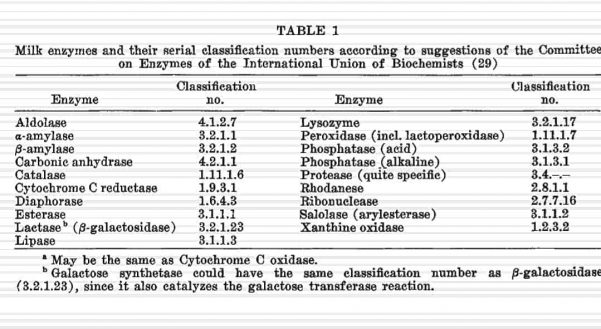
List of enzymes found in unpasteurized raw milk
Let’s get to know some of the enzymes that are found in unpasteurized raw milk.
Enzymes are destroyed in your gut as soon as they hit your stomach acid. If the milk is left alone, they aren’t destroyed, and they help influence the way the milk decomposes. This is useful if you make cheese.
Lactase
Lactase splits lactose into the two simple sugars glucose and galactose, which the body can then absorb through the intestinal wall. It is produced in the brush border of the small intestine, not secreted by the mammary gland. The cow makes no lactase, and none is present in freshly collected milk.
In short: lactase is not produced by the animal. It appears in raw milk only as fermentation begins – a process that pasteurization stops before it can develop.
Some people lose the ability to produce sufficient intestinal lactase as they age, which is what causes lactose intolerance. Trace amounts of lactase (technically beta-galactosidase) can accumulate in raw milk over time, but this is microbial in origin rather than native to the milk itself. As lactic acid bacteria colonize raw milk after collection and begin metabolizing lactose for energy, they produce beta-galactosidase intracellularly. When those cells lyse, some enzyme is released into the milk. This is the same biological process that, taken further, produces kefir and other fermented dairy products where beta-galactosidase concentrations are high enough to actually pre-digest a meaningful portion of the lactose. Refrigeration slows this bacterial activity considerably, so fresh raw milk destined for drinking contains only traces. Pasteurization eliminates both the bacteria and any enzyme they’ve produced.
The real-world implications are clearest in cheesemaking. The lactic acid bacteria that generate beta-galactosidase and other enzymes in raw milk are the same organisms responsible for the flavor complexity, proteolysis, and microbial terroir that define raw milk cheese. Research published in FEMS Microbiology Reviews (Quigley et al., 2013) and a 2023 study in Frontiers in Microbiology both document how the native LAB community in raw milk, impossible to replicate fully with added starter cultures, drives the distinctive character of traditionally made aged cheeses. Several protected designation of origin (PDO) cheeses in Europe require natural whey or raw milk cultures specifically because the indigenous microbial community is considered inseparable from the product’s identity. Pasteurization wipes that community out, and no amount of reintroduced starter culture fully recreates what was lost.
Alkaline Phosphatase
Phosphatase helps the body access phosphorus and calcium. Phosphatase hydrolyses uses water to break down phosphate esters, releasing phosphorus ions. Calcium absorption requires proper ratios of phosphorus and magnesium.
Phosphatase is completely destroyed at the lowest typical pasteurizing temperatures. Food processing companies test for the complete absence of phosphatase to determine if their pasteurization procedure was successful. Presumably, this process makes the absorption of phosphorus and calcium from milk difficult for our digestive systems.
Acid Phosphatase
Acid Phosphatase is akin to Alkaline Phosphatase. Acid Phosphatase works in environments with a lower pH than 7, while Alkaline Phosphatase works in environments higher than 7.
Amylase
Saliva and pancreatic juice contain Amylase to digest starch, glycogen, and other related carbohydrates. Raw milk also contains Amylase for the same reason. Amylase is commonly found in plants, as plants are mostly composed of carbohydrates. Amylase is rendered inactive by conventional milk processing (pasteurization and homogenization).
Catalase
Involved with waste management on the cellular level, catalase rids cells of hydrogen peroxide (H2O2), a chemical by-product of cellular metabolism. A strong oxidizer, H2O2 can wreak havoc in the cellular environment. Catalase quickly locks onto H2O2 and separates one oxygen atom from the molecule, producing oxygen and water. It appears to be rendered inactivate at temperatures above 158° F./70°C, well under the temperatures reached during pasteurization.
Lactoperoxidase
Lactoperoxidase is an antimicrobial enzyme native to raw milk that works in concert with two other naturally occurring compounds – thiocyanate ions and hydrogen peroxide – to form what is known as the lactoperoxidase system. This system inhibits bacterial growth and is thought to serve a protective function in the mammary gland and in freshly expressed milk. It is entirely separate from lysozyme, another antimicrobial enzyme present in milk, which works through a different mechanism by targeting bacterial cell walls directly.
Lactoperoxidase is relatively heat-resistant compared to most milk enzymes: roughly 50% of activity is retained in milk held at 158°F (70°C) for 20 minutes, but it is completely inactivated at 176°F (80°C) within 5 minutes. Because standard pasteurization temperatures typically fall between these thresholds, lactoperoxidase activity in commercially pasteurized milk varies depending on the exact heat treatment used. Food safety regulators use the complete absence of alkaline phosphatase, not lactoperoxidase, as the standard marker for successful pasteurization.
Lipase
Actually a class of water-soluble enzymes, lipases break down fats (triglycerides) into fatty acids, and improve utilization of lipids throughout the body. Disruption of the fat globules, as in homogenization, can lead to rancidity if lipase isn’t destroyed first. Pasteurization accomplishes this, increasing the milk’s shelf life. Lipase enzymes are normally inactive in raw milk until triggered by the proper pH in the digestive tract.
Xanthine Oxidase
One major protein component of the milk fat globule membrane surrounding lipid droplets in milk is Xanthine Oxidase.
Xanthine Oxidase (XOD) exhibits a broad substrate specificity including aldehydes, purines and pteridines. Furthermore, this enzyme reduces oxygen to generate superoxide, hydrogen peroxide and reactive oxygen species (ROS). It also reduces nitrite to yield reactive nitrogen species (RNS), such as peroxynitrite and nitric oxide. Owing to its ability to generate RNS and ROS, XOD might play an important role as an antimicrobial agent in the neonatal gut, thereby complementing endogenous enzyme of the intestinal epithelium. [Sigma Aldrich]
Gamma-glutamyl Transferase
γ-Glutamyl transferase activity is high in human colostrum, and although activity decreases thereafter, considerable amounts of enzyme are present in transitional and mature milk (Brinkley et al., 1975; Patii and Rangnekar, 1982).
It has been suggested that the enzyme is localized in the Golgi apparatus and that it plays a role in the endo- and/or exocytotic transport of proteins. [Science Direct]
Other Dairy Enzymes
Thanks to April for finding this chart!
Various links:
Science Direct: Milk Enzyme
Science Direct: Milk xanthine oxidase: Properties and physiological roles
Science Direct: Gamma Glutamyl Group
Journal of Dairy Science: Milk Enzymes, Their Role and Significance (pdf)
