
How Raw Milk Recalls Work: A Step-by-Step Guide to Food Safety in Action
A raw milk recall is not evidence that raw milk is dangerous. It is evidence that the food safety system is working.
Recalls are initiated because testing caught something — and testing catches things because licensed raw milk producers in states where retail sales are legal are among the most frequently tested food producers in the American food supply. When headlines generate alarm, the underlying story is usually the opposite: a surveillance system that detected a potential hazard, removed product from commerce, and guided a farm through remediation before resuming sales.
This article walks through that process in chronological order, from initial detection to the day a farm is cleared to resume operations.
Putting Recalls in Context: How Rare Is Raw Milk Illness?
Before examining the mechanics of a recall, some perspective is warranted. Between 2009 and 2014, an estimated 3.2% of the U.S. population — more than 10 million people — consumed raw milk regularly. That number has almost certainly grown since. Over that same period, the CDC attributed roughly 761 illnesses per year to raw milk, with 22 hospitalizations and no deaths attributable to raw milk alone.
That is a 0.007% illness rate — lower than the rate associated with leafy greens, chicken, or raw oysters. The outbreaks that do occur are almost always traceable to specific, identifiable lapses in biosecurity — not to any inherent danger in properly produced raw milk.
For comparison: in 2024, a Boar’s Head processing facility in Virginia was linked to 9 deaths and 57 hospitalizations across 18 states. Government inspectors documented 69 instances of non-compliance — mold, insects, blood pooling on floors, and rotten meat residue on walls. That industrial facility was held up as an example of food safety enforcement functioning as intended. The same logic applies when a licensed raw milk dairy triggers a recall: detection and response are the system working, not failing.
Social media periodically amplifies unverified claims about raw milk illness — one recurring example is a widely circulated image falsely depicting politicians sickened by raw milk, debunked by Snopes and rooted in a bout of illness that investigators could not link to raw milk at all. Consumers deserve accurate information, and the recall process described below is part of how accurate information gets produced.
The Legal Landscape: States Where Raw Milk Retail Sales Are Permitted
Raw milk retail sales are legal in roughly 30 states, with laws varying significantly by state — from full retail availability to farm-direct sales only. The raw milk law map provides a current, state-by-state breakdown of what is permitted where. Each state sets its own standards, but common requirements include herd health certification, routine bacterial testing of milk before release for sale, somatic cell count limits, facility sanitation standards, and labeling requirements. This pre-market testing infrastructure is what makes recalls possible — anomalies surface quickly because testing occurs continuously.
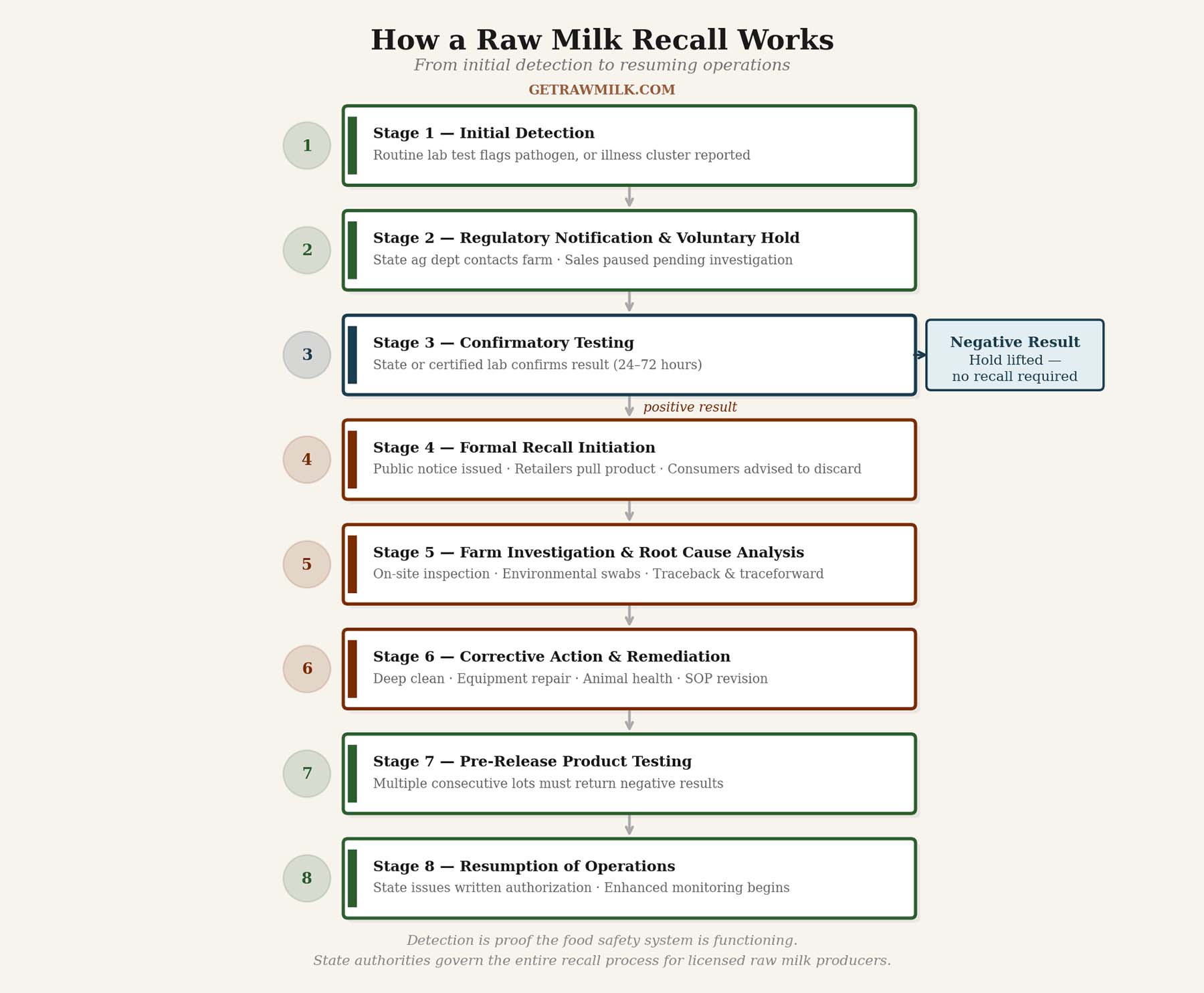
Stage 1: Initial Detection
Routine Testing Triggers a Flag
Most recalls begin not with a sick consumer, but with a laboratory result. State departments of agriculture require licensed raw milk producers to submit samples on a regular schedule — in many states, this means every batch or every few days. When a sample tests presumptively positive for a pathogen of concern — most commonly Listeria monocytogenes, Salmonella, E. coli O157:H7, or Campylobacter — the laboratory notifies the state regulatory agency immediately. This preliminary result is sufficient to initiate the recall process before confirmatory testing is complete.
Illness Cluster Investigations
In some cases detection begins epidemiologically. When public health officials identify a cluster of gastrointestinal illnesses with a common food exposure, they interview affected individuals. If raw milk from a specific dairy emerges as a common denominator, regulators initiate product testing and traceback investigations in parallel. Because raw milk is prohibited from interstate commerce under federal law, most recall actions remain within state jurisdiction, though the CDC and FDA may become involved when illness clusters cross state lines.
Stage 2: Regulatory Notification and Voluntary Hold
The Farm Is Notified
Upon a preliminary positive result or credible illness report, the state department of agriculture contacts the licensed dairy directly. In many cases, a farm with a robust on-farm testing program has already detected the issue before state contact occurs — which is precisely the outcome that proactive testing is designed to produce.
Voluntary Hold on Sales
Licensed producers in legal-sale states cooperate with a voluntary hold on sales and distribution while the situation is investigated. This is standard practice and reflects both consumer safety priorities and the producer’s long-term interests. Prompt voluntary action is consistently noted by regulators as a defining characteristic of licensed raw milk operations.
Confirmatory Testing
While the hold is in effect, the state laboratory or a certified private laboratory conducts confirmatory testing on retained samples. This typically takes 24 to 72 hours. A negative confirmatory result may end the process entirely with no recall required.
Stage 3: Formal Recall Initiation
If confirmatory testing returns a positive result, the state department of agriculture issues a formal recall order or works with the producer to announce a voluntary recall. The announcement identifies the dairy, the specific products and lot numbers affected, the pathogen detected, and instructions for consumers.
State agencies distribute notices through press releases, their own websites, direct outreach to retailers that carried the product, and increasingly through social media and email alert systems. Retail outlets are required to pull affected product and, where purchase records permit, contact customers directly.
Consumers who purchased recalled raw milk are advised to discard it or return it to the point of purchase, and to contact a healthcare provider if they are experiencing symptoms.
Stage 4: Farm Investigation and Root Cause Analysis
State agriculture inspectors conduct an on-farm investigation examining milking equipment sanitation, bulk tank condition and cleaning records, individual animal health, water source quality, employee hygiene practices, and environmental surfaces throughout the milking parlor and processing areas.
Environmental swabs are particularly important in Listeria investigations, as this pathogen can establish persistent reservoirs in processing environments and re-contaminate product after cleaning. Regulators conduct parallel traceback and traceforward investigations — tracing the contamination to its source while also mapping where affected product was distributed and what remains in commerce.
Stage 5: Corrective Action and Remediation
Before a farm can resume sales, regulators require a written corrective action plan (CAP) identifying the probable root cause and detailing specific remediation steps. Common corrective actions include deep cleaning and sanitizing all contact surfaces, replacing or repairing equipment identified as a contamination vector, enhanced water source testing, culling of animals that tested positive, and revision of sanitation procedures.
For Listeria in particular, regulatory agencies typically require multiple rounds of negative environmental swabs — often over several weeks — before authorizing a production restart.
Stage 6: Pre-Release Testing and Resumption of Operations
Before the farm is permitted to resume sales, regulators require enhanced product testing — typically multiple consecutive lots, all returning negative results. This documents both the effectiveness of corrective actions and the farm’s return to compliance.
The state department of agriculture then issues formal written authorization to resume sales. In most states, this authorization is announced through the same public channels as the original recall, closing the loop for consumers. The farm then enters a period of enhanced regulatory monitoring: more frequent inspections, additional required testing, and closer review of records.
The Role of the FDA and CDC
Because raw milk cannot legally cross state lines, recalls are primarily a state-level function. That distinction matters more now than it did a few years ago.
The Trump administration’s restructuring of the Department of Health and Human Services has reduced the federal food safety apparatus in meaningful ways. In April 2025, the FDA suspended its proficiency testing program for Grade A milk — an internal quality control program that verified lab accuracy for dairy testing — due to workforce cuts. As of July 2025, the CDC reduced its FoodNet surveillance network from eight pathogens to two (Salmonella and STEC), making active federal monitoring for Listeria, Campylobacter, and others optional rather than required. PulseNet — the CDC’s whole genome sequencing network for linking illness cases — remains operational, though its long-term funding is uncertain.
None of these federal changes affect the state-level testing requirements that govern licensed raw milk producers. Raw milk produced for direct human consumption is regulated and tested by state authorities, not the FDA. State laws mandating routine testing remain fully in effect and will continue to be enforced by state-approved labs regardless of what happens at the federal level.
The practical effect for raw milk recalls: state departments of agriculture remain the primary enforcement authority. The FDA may still become involved if a raw milk product is suspected of entering interstate commerce illegally, but the core recall machinery — testing, notification, corrective action, authorization to resume sales — runs entirely through the states.
How Recalls Demonstrate the Strength of Raw Milk Oversight
Recalls happen because testing found something. Testing found something because testing occurs — routinely, rigorously, and at meaningful frequency. The raw milk producers operating under state licensing in legal-sale states are among the most tested food producers in the American food system on a per-unit basis.
That sequence — detection, response, remediation, verification, resumption — is what a functioning food safety system looks like. It is the same sequence described approvingly when the Boar’s Head outbreak was eventually contained. The difference is scale: that outbreak killed nine people. Most raw milk recalls involve zero confirmed illnesses.
On-Farm Testing: Stopping Problems Before They Leave the Farm
The most effective intervention in any raw milk food safety event is the test that catches a problem before a single bottle enters commerce. A recall is, by definition, a response to contamination that has already been distributed. Routine on-farm testing — conducted by the producer as part of a proactive food safety program — is the opportunity to avoid a recall entirely.
Producers monitoring bacterial counts regularly, tracking trends over time, and testing for pathogens on a consistent schedule are far more likely to detect an emerging issue at the farm level. The problem is identified, the lot is held or discarded, corrective action is taken, and consumers are never exposed. As RAWMI trainer Mark McAfee puts it: the goal is “no surprises.”
The Raw Milk Institute (RAWMI) Model
The Raw Milk Institute (RAWMI) has formalized this proactive approach through its Common Standards and Listed producer program. Developed by an international group of medical doctors, epidemiologists, food safety scientists, and veterinarians, the program centers on three pillars: farmer training and mentoring, a farm-specific Risk Analysis and Management Plan (RAMP), and rigorous ongoing testing. RAWMI offers a free online safety course covering risk management from grass to glass.
Under RAWMI standards, listed farms target coliform counts below 10 per milliliter and Standard Plate Count below 5,000 per milliliter — thresholds that reflect a genuine commitment to low-risk production, not merely minimum compliance. Farms test at least monthly, report results transparently, and use trend data as an early warning system. RAWMI has published a detailed overview of on-site raw milk testing methods, and producers can watch a farmer test raw goat milk on the farm to see the process in action.
A farm actively monitoring its own milk is likely to detect an upward shift in bacterial counts — a signal that something has changed in the environment, the herd, or the process — well before that shift escalates into a confirmed pathogen event. That early detection is what keeps a potential problem from becoming a public recall. Consumers can search for RAWMI-listed producers to find farms committed to this standard.
What Consumers Can Do
- Sign up for recall alerts from their state department of agriculture. Most states offer email or text notifications for food safety recalls.
- Keep purchase records — producer name, lot number, and sell-by date. This information is essential if a recall is announced.
- Follow the dairy’s own channels. Many licensed raw milk producers communicate directly with customers via email lists or social media, including during recall events.
- Know the symptoms of common foodborne pathogens — diarrhea, vomiting, fever, abdominal cramping — and contact a healthcare provider promptly if symptoms develop after consumption.
- Seek out RAWMI-listed producers where possible. These farms have committed to proactive, transparent testing specifically designed to catch problems before product reaches consumers.
- Understand household risk profiles. Regulatory agencies consistently identify pregnant women, young children, the elderly, and immunocompromised individuals as groups warranting particular consideration.
Transparency Is the Foundation of Trust
From the moment a test flags a concern to the day a farm is authorized to resume sales, the raw milk recall process is documented, regulated, and oriented toward a single outcome: safe food reaching consumers who have made an informed choice to purchase it. Detection is not the story — it is the precondition for the story going well.
